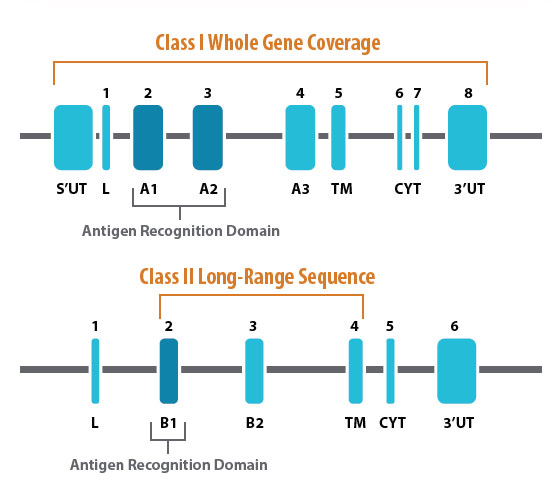
Next Generation Sequencing (NGS) technology increases the resolution of the HLA typing, while offering a more economical genotyping approach compared to conventional sequencing methods. Labcorp offers NGS typing of all major HLA loci in both ARD/(G-Group) and long read formats, providing options to achieve better resolution, turn-around time, and cost.1
Alternative short read technologies may suffer from typing ambiguities when heterozygotes include similar alleles. Labcorp’s technology allows for the rapid resolution of these G-Group ambiguities.
For the highest resolution needs, Labcorp offers third generation long-read sequencing. For HLA class I loci, this assay sequences the entire gene in a single read, eliminating nearly all ambiguities. For Class II loci, the long-read assay covers exons 2, 3 and 4, as well as the intervening intron sequences. Labcorp’s long-read NGS assay provides up to four (4) field resolution and may be reported as an expanded result containing variants within the coding region and polymorphic differences in the non-coding region. All alleles containing differences in gene expression are captured in the final result.
Labcorp also offers other sequencing methods to expedite turnaround time. For more information please contact your Labcorp representative or call customer service at 800-533-1037 or by email at HLACS@labcorp.com.